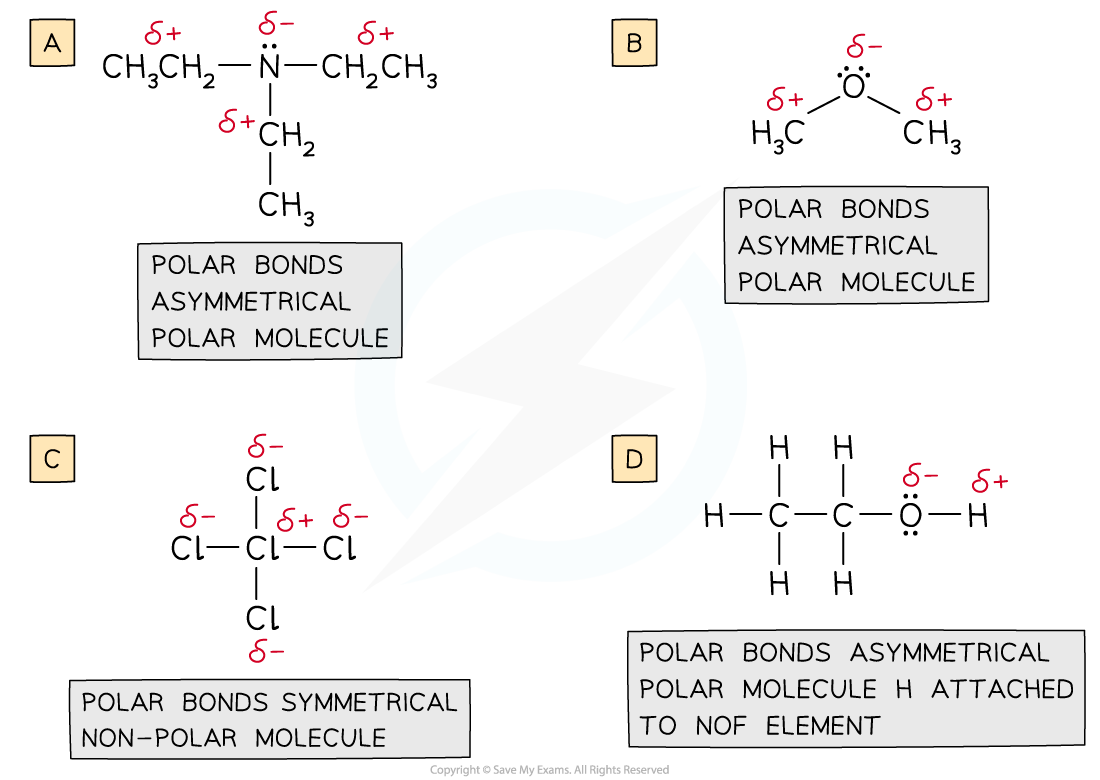
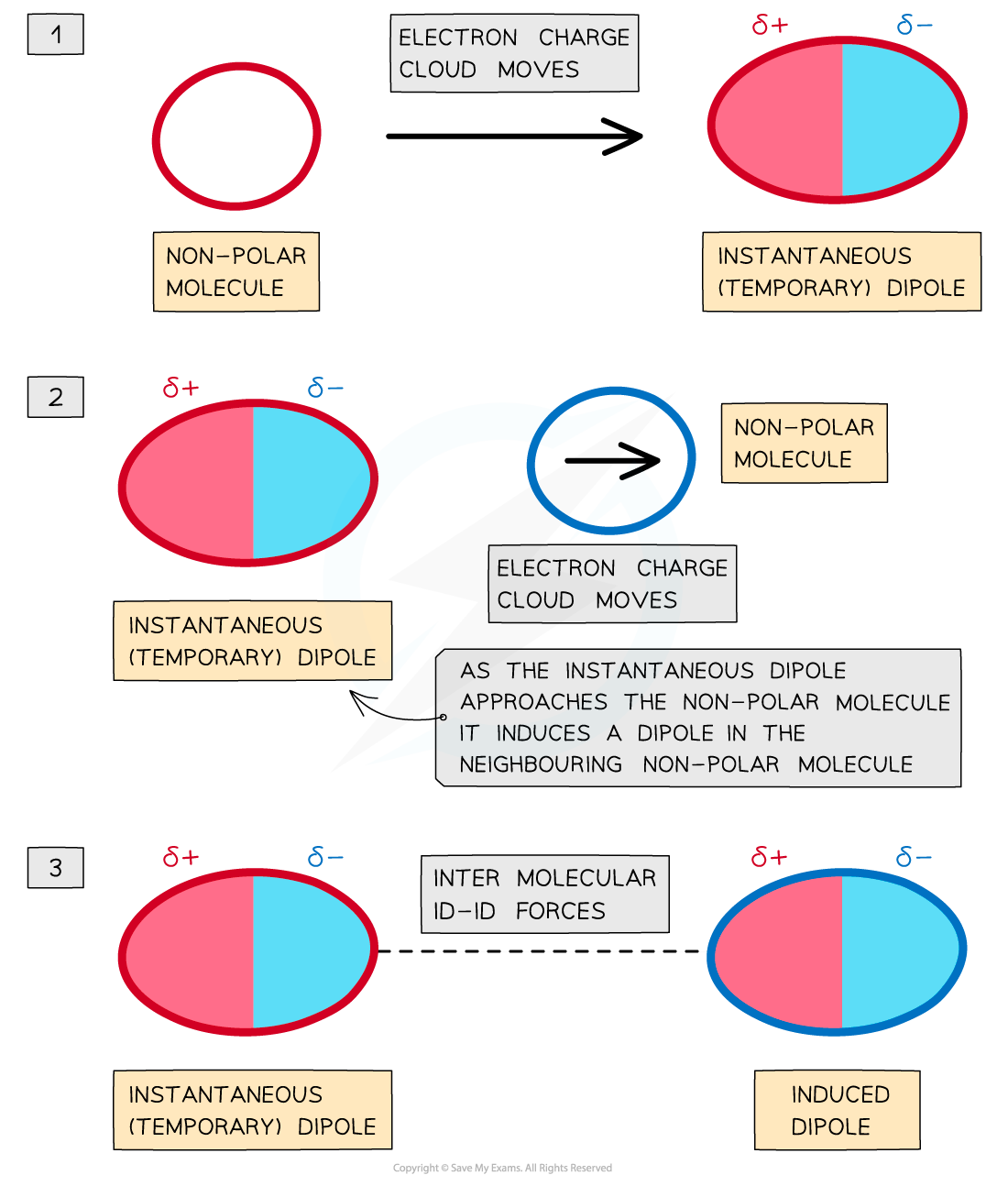
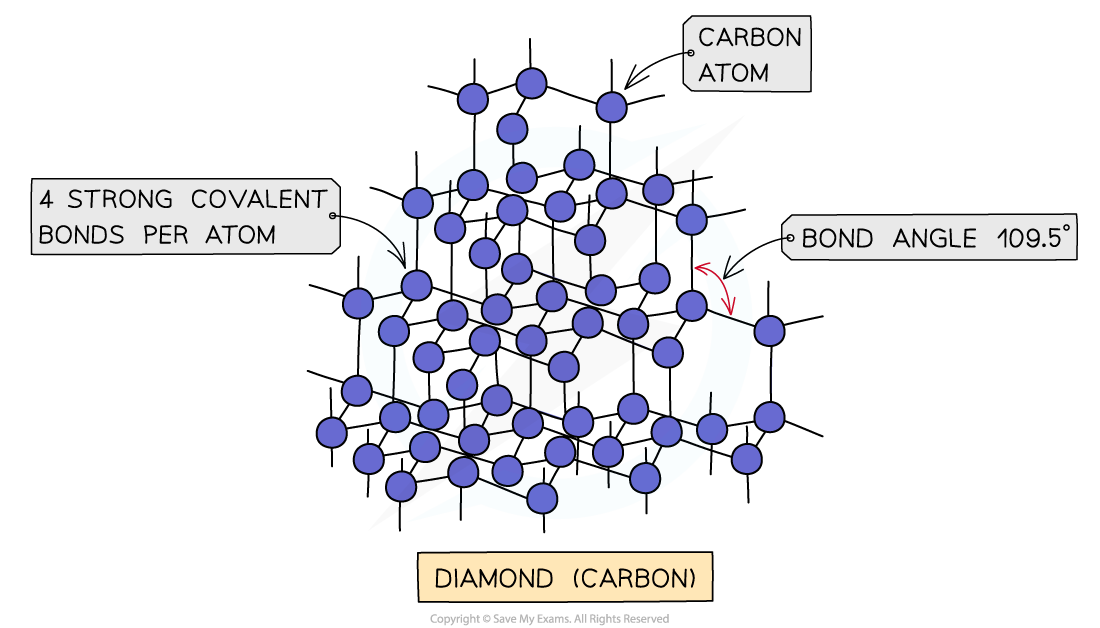
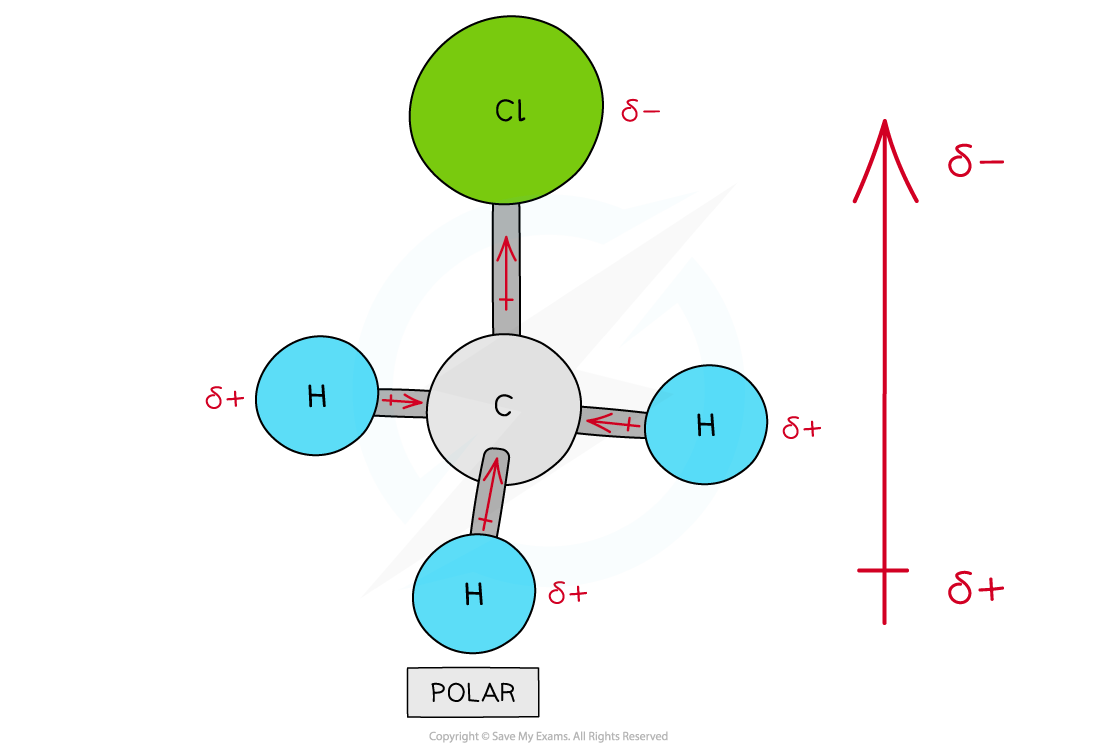
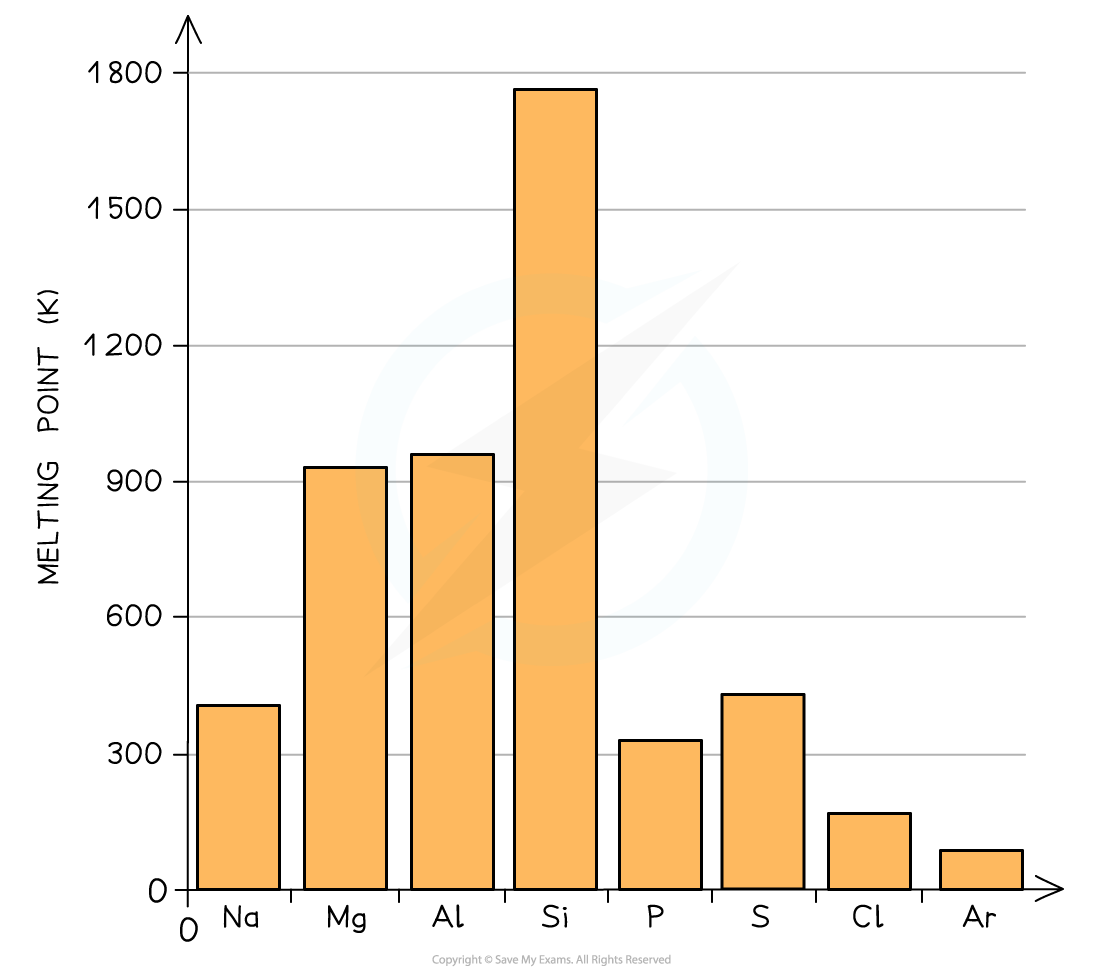IB DP Chemistry: SL复习笔记4.3.6 Alloys & their Properties
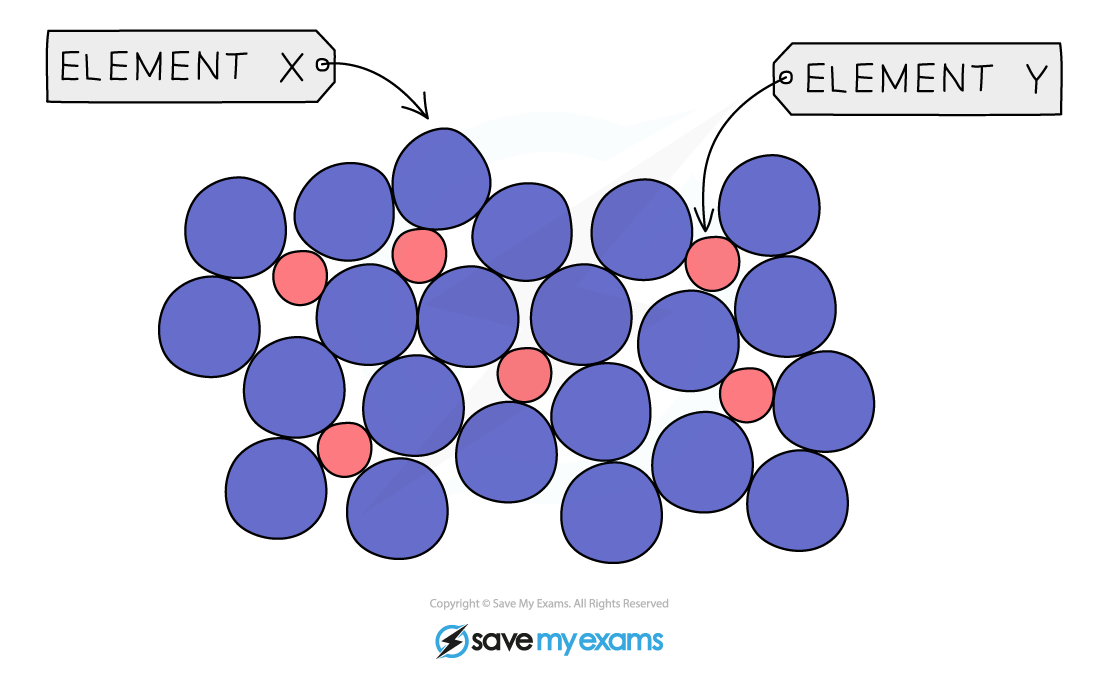
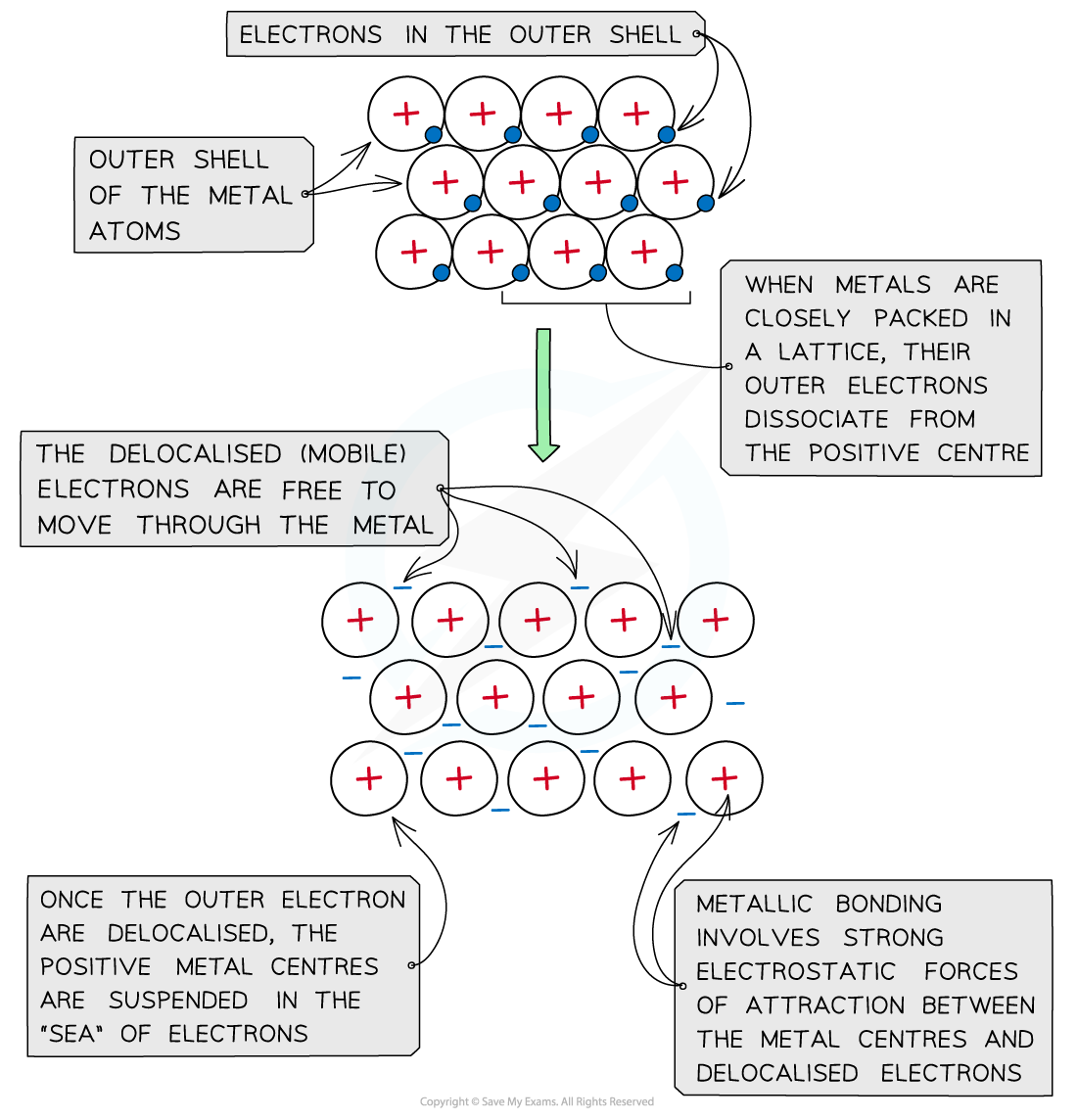
Alloys Alloys are mixtures of metals, where the metals are mixed together physically but are not chemically combined They can also be made from metals mixed with nonmetals such as carbon Ions of th...