- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 021-63526630
AQA A Level Chemistry复习笔记1.5.2 Bond Polarity
Electronegativity
- Electronegativity is the power of an atom to attract the pair of electrons in a covalent bond towards itself
- The electron distribution in a covalent bond between elements with different electronegativities will be unsymmetrical
- This phenomenon arises from the positive nucleus’s ability to attract the negatively charged electrons, in the outer shells, towards itself
- The Pauling scale is used to assign a value of electronegativity for each atom
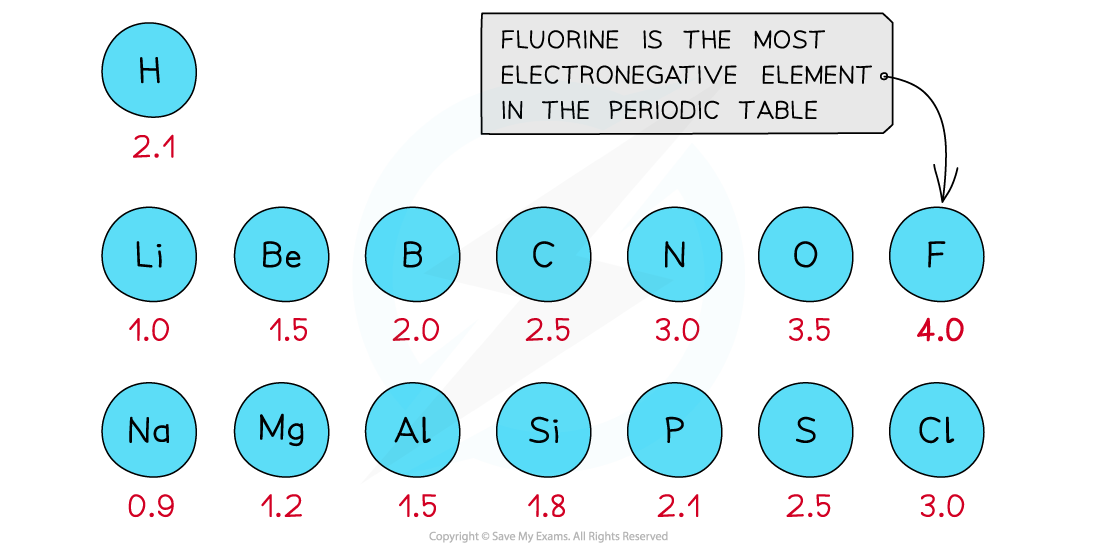
First three rows of the periodic table showing electronegativity values
- Fluorine is the most electronegative atom on the Periodic Table, with a value of 4.0 on the Pauling Scale
- It is best at attracting electron density towards itself when covalently bonded to another atom
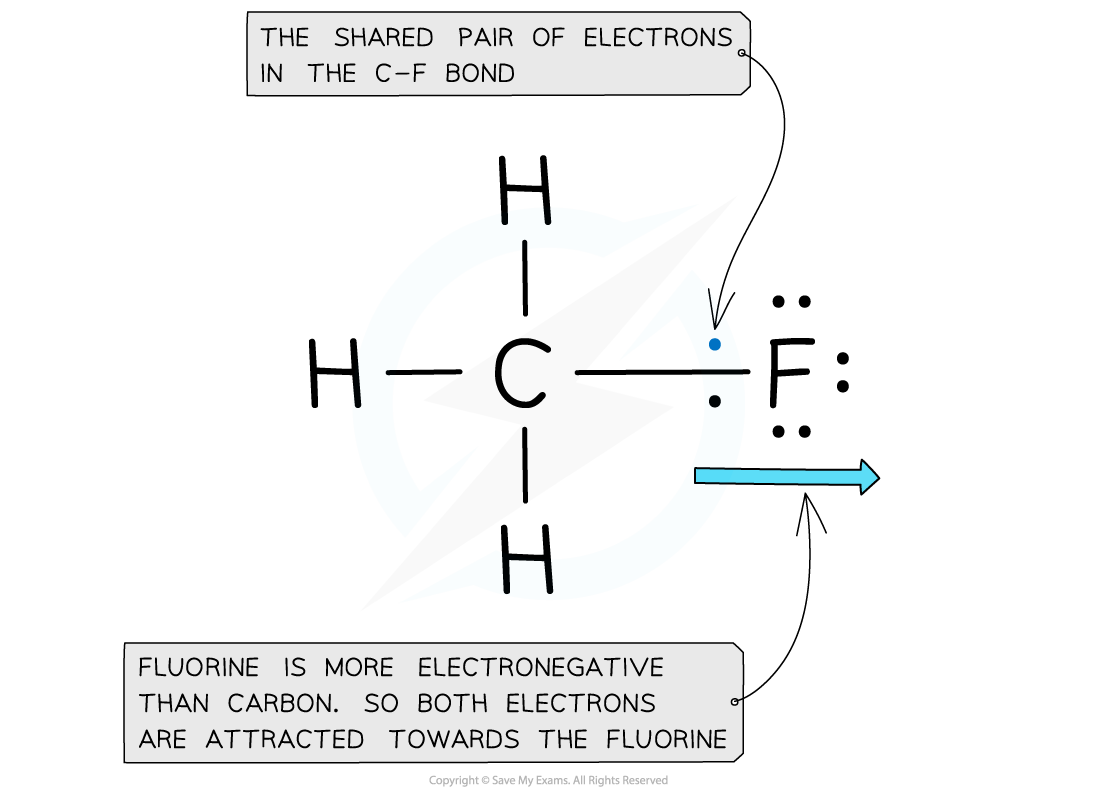
Electron distribution in the C-F bond of fluoromethane
Nuclear charge
- Attraction exists between the positively charged protons in the nucleus and negatively charged electrons found in the energy levels of an atom
- An increase in the number of protons leads to an increase in nuclear attraction for the electrons in the outer shells
- Therefore, an increased nuclear charge results in an increased electronegativity
Atomic radius
- The atomic radius is the distance between the nucleus and electrons in the outermost shell
- Electrons closer to the nucleus are more strongly attracted towards its positive nucleus
- Those electrons further away from the nucleus are less strongly attracted towards the nucleus
- Therefore, an increased atomic radius results in a decreased electronegativity
Shielding
- Filled energy levels can shield (mask) the effect of the nuclear charge causing the outer electrons to be less attracted to the nucleus
- Therefore, the addition of extra shells and subshells in an atom will cause the outer electrons to experience less of the attractive force of the nucleus
- Sodium (period 3, group 1) has higher electronegativity than caesium (period 6, group 1) as it has fewer shells and therefore the outer electrons experience less shielding than in caesium
- Thus, an increased number of inner shells and subshells will result in a decreased electronegativity
Trends in Electronegativity
- Electronegativity varies across periods and down the groups of the periodic table
Down a group
- There is a decrease in electronegativity going down the group
- The nuclear charge increases as more protons are being added to the nucleus
- However, each element has an extra filled electron shell, which increases shielding
- The addition of the extra shells increases the distance between the nucleus and the outer electrons resulting in larger atomic radii
- Overall, there is decrease in attraction between the nucleus and outer bonding electrons
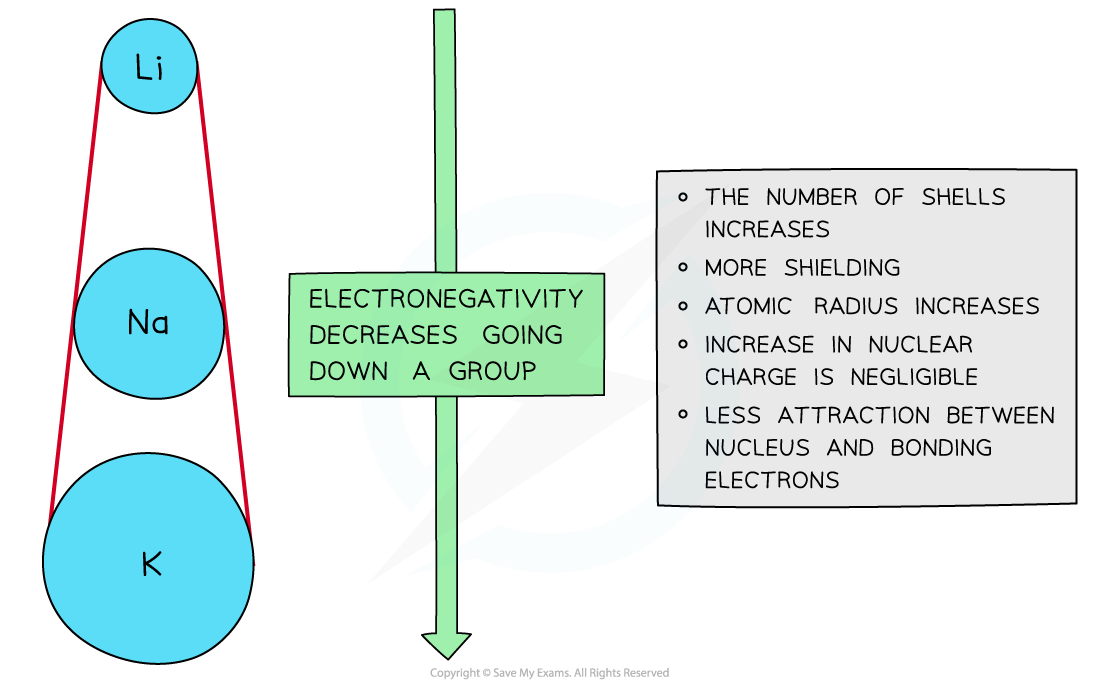
Electronegativity decreases going down the groups of the periodic table
Across a period
- Electronegativity increases across a period
- The nuclear charge increases with the addition of protons to the nucleus
- Shielding remains relatively constant across the period as no new shells are being added to the atoms
- The nucleus has an increasingly strong attraction for the bonding pair of electrons of atoms across the period of the periodic table
- This results in smaller atomic radii
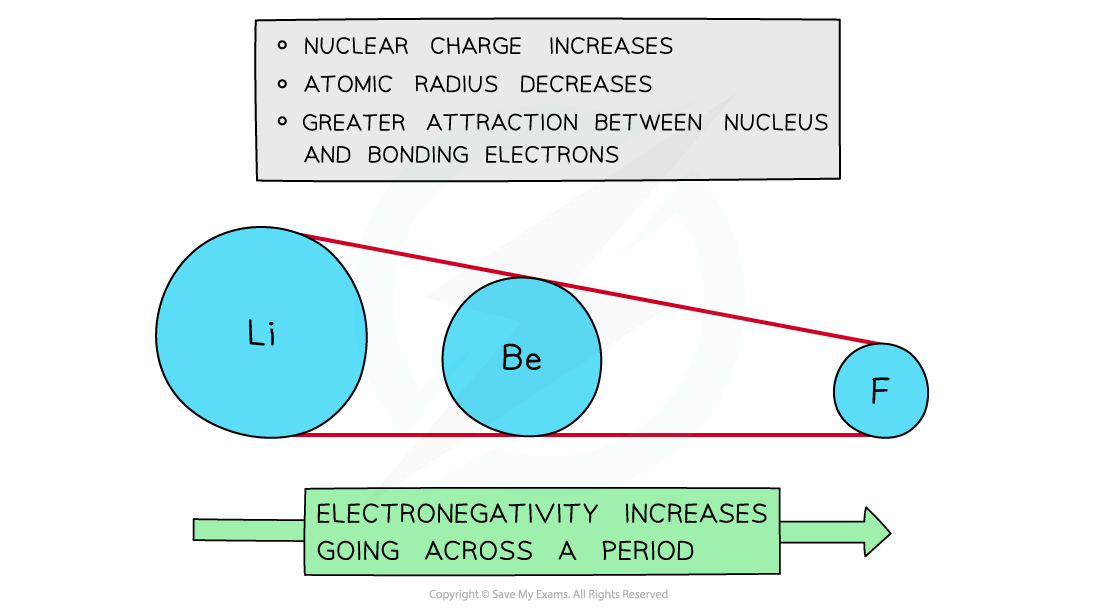
Electronegativity increases going across the periods of the Periodic Table
Exam Tip
Remember the general trend is an increase in electronegativity towards the top right of the periodic table.Fluorine is the most electronegative element in the periodic table.
转载自savemyexams
以上就是关于【AQA A Level Chemistry复习笔记1.5.2 Bond Polarity】的解答,如需了解学校/赛事/课程动态,可至翰林教育官网获取更多信息。
往期文章阅读推荐:
全网破防!ALevel CIE数学M1疑似错题?经济P2难度飙升?5月6日大考考情分析必看!
A-Level CIE就大规模泄题发布最严处罚!哪些考生必须重考?你的成绩怎么办?

最新发布
© 2026. All Rights Reserved. 沪ICP备2023009024号-1









