- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 021-63526630
AQA A Level Chemistry复习笔记1.1.7 Ionisation Energy: Trends & Evidence
Ionisation Energies: Equations
- The second ionisation energy (IE2) is the energy required to remove the second mole of electrons from each +1 ion in a mole of gaseous +1 ions, to form one mole of +2 ions
- The third ionisation energy (IE3) is the energy required to remove the third mole of electrons from each +2 ion in a mole of gaseous +2 ions, to form one mole of +3 ions
- And so on...
- The electrons from an atom can be continued to be removed until only the nucleus is left
- This sequence of ionisation energies is called successive ionisation energies
Successive Ionisation Energies of Beryllium Table
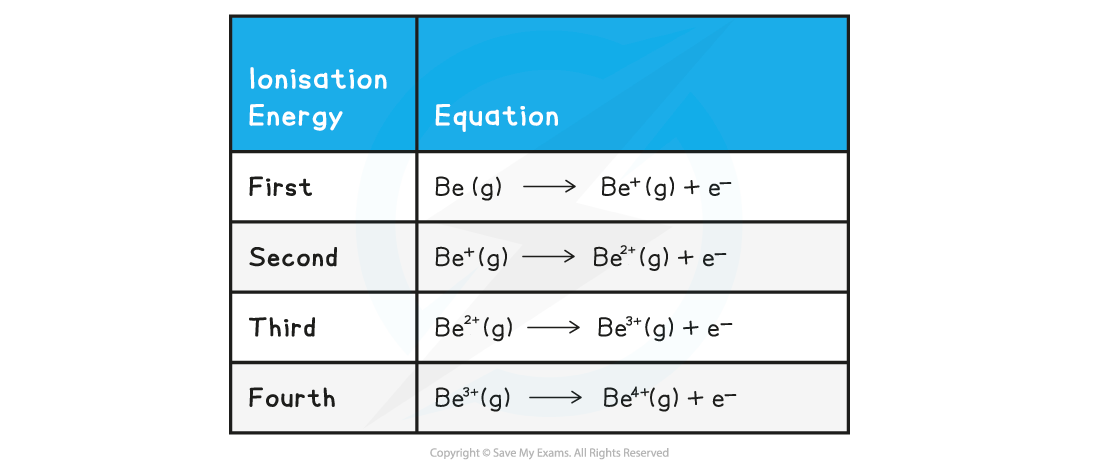
Exam Tip
Remember that equations to represent ionisation energies must have gaseous (g) state symbols for the atoms and ions but not for the electrons.You will lose the mark in your exam if you do not include the state symbols, even if the question does not specify for you to include them.
Successive Ionisation Energies
Successive ionisation energies of an element
- The successive ionisation energies of an element increase
- This is because once you have removed the outer electron from an atom, you have formed a positive ion
- Removing an electron from a positive ion is more difficult than from a neutral atom
- As more electrons are removed, the attractive forces increase due to decreasing shielding and an increase in the proton to electron ratio
- The increase in ionisation energy, however, is not constant and is dependent on the atom's electronic configuration
- Taking calcium as an example:
Ionisation Energies of Calcium Table
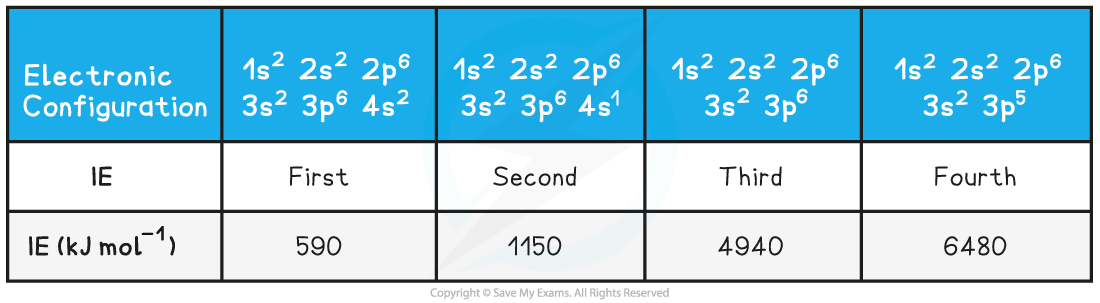
- The first electron removed has a low IE1 as it is easily removed from the atom due to the spin-pair repulsion of the electrons in the 4s orbital
- The second electron is more difficult to remove than the first electron as there is no spin-pair repulsion
- The third electron is much more difficult to remove than the second one corresponding to the fact that the third electron is in a principal quantum shell which is closer to the nucleus (3p)
- Removal of the fourth electron is more difficult as the orbital is no longer full, and there is less spin-pair repulsion
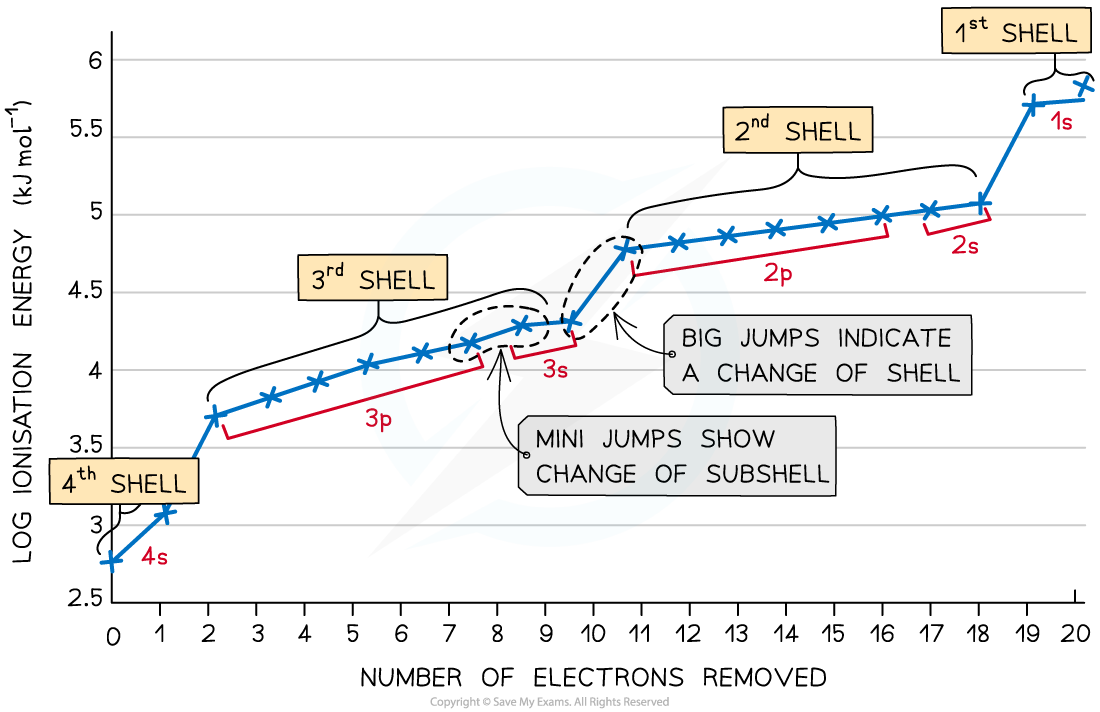
- The graph shows there is a large increase in successive ionisation energy as the electrons are being removed from an increasingly positive ion
- The big jumps on the graph show the change of shell and the small jumps are the change of subshell
Exam Tip
It gets more difficult to remove electrons from principal quantum shells that get closer to the nucleus, as there is less shielding and an increase in attractive forces between the electrons and nuclear charge.Be careful with how you interpret successive ionisation energy graphs, especially if you are not given every successive ionisation energy and are just shown part of the graph - you should count the electrons from left to right!It is a good idea to label the shells and subshells on ionisation energy graphs in an exam, so that you do not make the mistake of reading the graph backwards.
- Successive ionisation data can be used to:
- Predict or confirm the simple electronic configuration of elements
- Confirm the number of electrons in the outer shell of an element
- Deduce the Group an element belongs to in the Periodic Table
- By analyzing where the large jumps appear and the number of electrons removed when these large jumps occur, the electron configuration of an atom can be determined
- Na, Mg and Al will be used as examples to deduce the electronic configuration and positions of elements in the Periodic Table using their successive ionisation energies
Successive Ionisation Energies Table

Sodium
- For sodium, there is a huge jump from the first to the second ionisation energy, indicating that it is much easier to remove the first electron than the second
- Therefore, the first electron to be removed must be the last electron in the valence shell thus Na belongs to group I
- The large jump corresponds to moving from the 3s to the full 2p subshellNa 1s2 2s2 2p6 3s1
Magnesium
- There is a huge increase from the second to the third ionisation energy, indicating that it is far easier to remove the first two electrons than the third
- Therefore the valence shell must contain only two electrons indicating that magnesium belongs to group II
- The large jump corresponds to moving from the 3s to the full 2p subshellMg 1s2 2s2 2p6 3s2
Aluminium
- There is a huge increase from the third to the fourth ionisation energy, indicating that it is far easier to remove the first three electrons than the fourth
- The 3p electron and 3s electrons are relatively easy to remove compared with the 2p electrons which are located closer to the nucleus and experience greater nuclear charge
- The large jump corresponds to moving from the third shell to the second shellAl 1s2 2s2 2p6 3s2 3p1
转载自savemyexams
以上就是关于【AQA A Level Chemistry复习笔记1.1.7 Ionisation Energy: Trends & Evidence】的解答,如需了解学校/赛事/课程动态,可至翰林教育官网获取更多信息。
往期文章阅读推荐:
全网破防!ALevel CIE数学M1疑似错题?经济P2难度飙升?5月6日大考考情分析必看!
A-Level CIE就大规模泄题发布最严处罚!哪些考生必须重考?你的成绩怎么办?

最新发布
© 2026. All Rights Reserved. 沪ICP备2023009024号-1









