- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 021-63526630
AQA A Level Physics复习笔记2.4.1 The Electronvolt
The Electronvolt
- The electronvolt is a unit which is commonly used to express very small energies
- This is because quantum energies tend to be much smaller than 1 Joule
- The electronvolt is derived from the definition of potential difference:
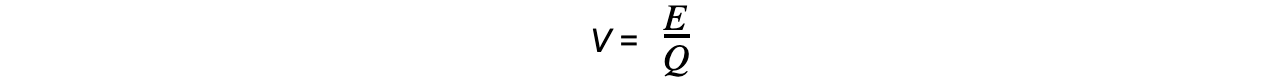
- When an electron travels through a potential difference, energy is transferred between two points in a circuit, or electric field
- If an electron, with a charge of 1.6 × 10-19 C, travels through a potential difference of 1 V, the energy transferred is equal to:
E = QV = 1.6 × 10-19 C × 1 V = 1.6 × 10-19 J
- Therefore, an electronvolt is defined as:
The energy gained by an electron travelling through a potential difference of one volt
1 eV = 1.6 × 10-19 J
Relation to kinetic energy
- When a charged particle is accelerated through a potential difference, it gains kinetic energy
- If an electron accelerates from rest, an electronvolt is equal to the kinetic energy gained:
eV = ½ mv2
- Rearranging the equation gives the speed of the electron:
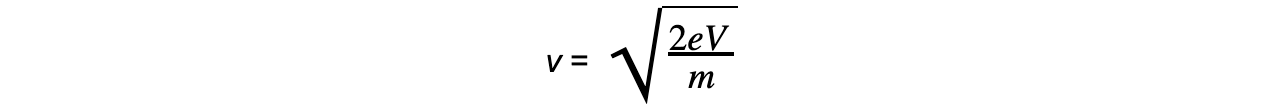
Worked Example
Show that the photon energy of light with wavelength 700nm is about 1.8 eV.
Step 1: Write the equations for wave speed and photon energy


Step 2: Calculate the photon energy in Joules
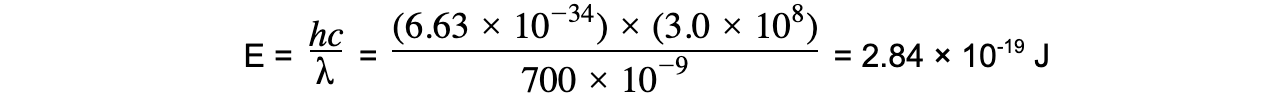
Step 3: Convert the photon energy into electronvolts
1 eV = 1.6 × 10-19 J J → eV: divide by 1.6 × 10-19
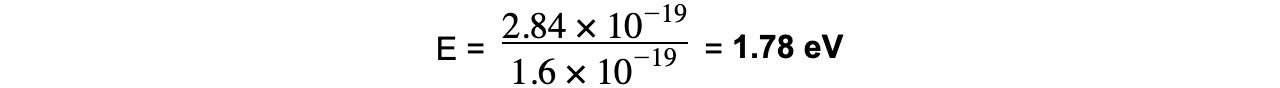
Exam Tip
- To convert between eV and J:
- eV → J: multiply by 1.6 × 10-19
- J → eV: divide by 1.6 × 10-19
转载自savemyexams
以上就是关于【AQA A Level Physics复习笔记2.4.1 The Electronvolt】的解答,如需了解学校/赛事/课程动态,可至翰林教育官网获取更多信息。
往期文章阅读推荐:
全网破防!ALevel CIE数学M1疑似错题?经济P2难度飙升?5月6日大考考情分析必看!
A-Level CIE就大规模泄题发布最严处罚!哪些考生必须重考?你的成绩怎么办?

最新发布
© 2026. All Rights Reserved. 沪ICP备2023009024号-1









